A Novel Cre Recombinase-Mediated In Vivo Minicircle DNA (CRIM) Vaccine Provides Partial Protection against Newcastle Disease Virus
Authors
Yanlong Jiang a, Xing Gao a, Ke Xu a, Jianzhong Wang a, Haibin Huang a, Chunwei Shi a, Wentao Yang a, Yuanhuan Kang a, Roy Curtiss III a, Guilian Yang b, Chunfeng Wanga a. (2019)
a. College of Animal Science and Technology, Jilin Provincial Engineering Research Center of Animal Probiotics, Key Lab of Animal Production, Product Quality and Security, Ministry of Education, Jilin Agricultural University, Changchun, China
b. Department of Infectious Disease and Immunology, College of Veterinary Medicine, University of Florida, Gainesville, Florida, USA
Introduction
As a paramyxovirus, the Newcastle disease virus (NDV), poses a severe threat for poultry production. The dominant strain in China is the genotype VII NDV, which has resulted in significant economic loss for poultry production. One of the major problems is the immunization route which limits the application of these vaccines in the field. Moreover, plasmid size could decrease the efficiency of plasmid translocation from the cytoplasm to the nucleus. To overcome this disadvantage, a newly defined minicircle DNA (mcDNA) containing only the eukaryotic promoter and gene of interest has attracted attention. The parental plasmid could be transformed into mcDNA and a miniplasmid by site-specific recombinases such as the Cre recombinase. Being much smaller, mcDNA could dramatically increase the efficiency of translocation into the nucleus, resulting in high expression levels. The regulated-delayed-lysis Salmonella strain, genetically engineered to lyse gradually in vivo after immunization, provides an attractive DNA vaccine delivery platform without an antibiotic selection marker. Salmonella lysis is based on the asd and murA genes, responsible for complete bacterial cell wall synthesis.
Objective
In this study, we built up a novel Cre recombinase-mediated in vivo mcDNA (CRIM) platform for the production of mcDNA in vivo based on the presence of Cre recombinase driven by the PGK promoter. The plasmid could replicate as a complete parental plasmid in vitro and then transform into mcDNA in vivo by itself. Then, we used this CRIM platform to express the HN gene of genotype VII NDV and evaluated the protective effects of the mcDNA when delivered by regulated-delayed-lysis Salmonella.
Material & methods
Synthesis of EGFP in vivo by pYL19 and pYL46 delivered by the Salmonella vector. To determine whether our novel CRIM platform works in vivo, we first transformed both the pYL19 and pYL46 plasmids into competent X11218 cells. 30 days old laying hens were orally inoculated with approximately 1 x109 CFU of X11218 harboring the empty vector pYA4545, pYL19, or pYL46 in 100 µl of saline, with three birds per group. At day 7 postinoculation, all the chickens were necropsied, and the livers were collected and subjected to in vivo imaging (Newton 7.0, Vilber Lourmat); then, the fluorescence intensities of the synthesized EGFP were determined. All experimental procedures and animal management procedures complied with the requirements of the Animal Care and Ethics Committees of Jilin Agriculture University, China.
Results
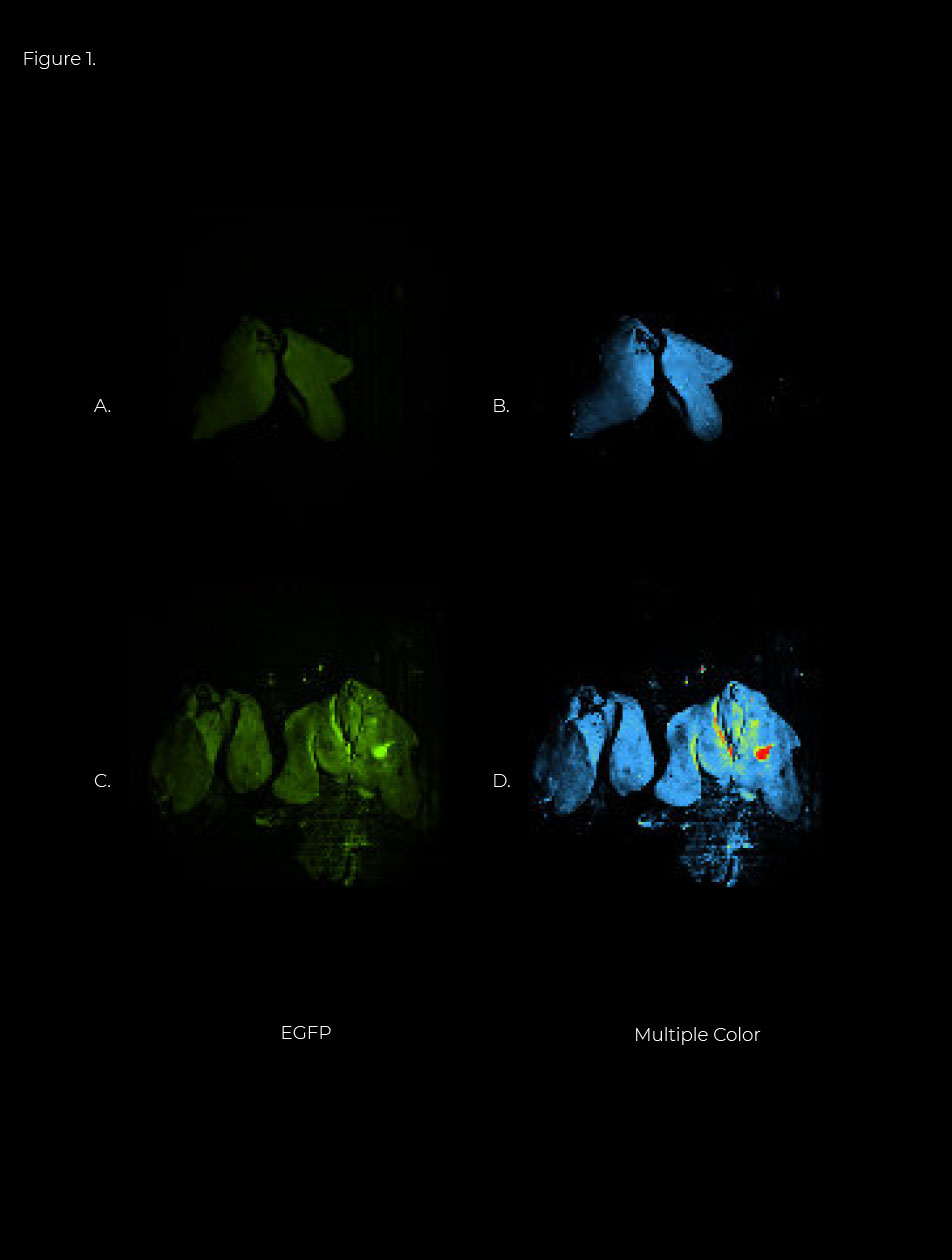
In vivo visualization of the increased production of EGFP by pYL46 delivered by Salmonella in chickens. To determine whether the increased production of EGFP by pYL46 could be observed in vivo, chickens were orally inoculated with the Salmonella strain X11218 harboring pYA4545, pYL19, or pYL46, and liver samples were collected at day 7 postinoculation and subjected to in vivo imaging using the Newton 7.0 (Vilber Lourmat, France). Compared with levels in livers from the X 11218(pYA4545) group (Figure 1.A and B) and X11218(pYL19) group (Fig. 2 D and E, left), a distinct increase in fluorescence intensity was observed in the X11218 (pYL46) group (Figure 1. D and E, right). The results showed that the EGFP/chicken GAPDH (cGAPDH) ratio in the X11218(pYL46) group was significantly higher than that in the X11218(pYL19) group (P <0.001). All of the results (ref. to publication) demonstrated that the Salmonella strain could deliver the EGFP gene cassette into chickens more efficiently with our CRIM vector than with a conventional plasmid.
Figure 1. Increased delivery of EGFP to chicken livers by Salmonella harboring pYL46. Chickens were orally inoculated with 1 x109 CFU of recombinant Salmonella X11218 carrying pYA4545, pYL19, or pYL46. The livers were collected at day 7 postinoculation from chickens immunized with X11218 (pYA4545) (A and B), X11218 (pYL19) (C, and D, left), and X11218 (pYL46) (C and D, right).
Results
In the present report, we describe the spontaneous transformation of a plasmid to mcDNA in vivo. The purpose of this study was to design a novel in vivo release platform for mcDNA production delivered by Salmonella for veterinary vaccine studies. We constructed a novel plasmid containing both the Cre recombinase under the phosphoglycerate kinase (PGK) promoter and recombinant lox66 and lox71 sites located outside the cytomegalovirus (CMV) expression cassette. The strictly controlled synthesis of Cre recombinase in vivo maintained the complete form of the plasmid in vitro, whereas the in vivo production of Cre transformed the parental plasmid to mcDNA after transfection. This newly designed Cre recombinase-mediated in vivo mcDNA platform, named CRIM, significantly increased the nuclear entry of mcDNA, followed by increased production of mRNA and protein, using enhanced green fluorescent protein (EGFP) as a model. In combination with our previous regulated-delayed-lysis Salmonella vector, the novel CRIM vaccine provided significant protection against wild-type NDV challenge in a chicken model, indicating that our novel Salmonella-CRIM technology could provide a novel platform for poultry vaccine development.