In vivo antitumor activity of locoregional
211At-trastuzumab in the treatment of peritoneal metastasis of gastric cancer in mice
Authors
Huizi Keiko Li1,2,3, Yukie Morokoshi1, Kotaro Nagatsu4, Tadashi Kamada2,5, and Sumitaka Hasegawa1 (2017)
1. Radiation and Cancer Biology Team, National Institutes for Quantum and Radiological Science and Technology
2. Graduate School of Medical and Pharmaceutical Sciences, Chiba University, Chiba
3. Japan Society for the Promotion of Science, Tokyo
4. Targetry and Target Chemistry Team
5. Clinical Research Cluster, National Institutes for Quantum and Radiological Science and Technology, Chiba, Japan
Introduction
Gastric cancer (GC) is one of the leading causes of cancer-related death worldwide. Recent advances, identified reliable predictive biomarkers such as human epidermal growth factor receptor (HER2), thus introducing molecular targeted drug therapy. GC’s HER2-positive play an important role in the development and progression of certain aggressive types of breast cancer. Trastuzumab, an anti-HER2 mAb (monoclonal antibody), has been used previously as a targeted therapy for HER2-positive GC. It induces an immune-mediated response that causes internalization and downregulation of HER2 and yields excellent treatment outcomes. Radioimmunotherapy (RIT) is a targeted radioisotope treatment method that uses an antibody as a carrier of therapeutic radioisotopes. The selective targeting of radioisotopes to the tumor using a radiolabeled cancer-specific antibody enables the delivery of a high dose of radiation directly to cancer cells while minimizing the exposure of normal cells. 211At is of particular interest because it emits highly cytotoxic α -particles that can kill a target cell, making it one of the most potent cell-killing agents available. Astatine-211 is therefore particularly suited to the targeted killing of disseminated or micrometastatic solid tumors that are usually resistant.
Objective
We hypothesized that astatine-211-labeled trastuzumab (211At-trastuzumab or 211At-tras) emitting highly cytotoxic α -particles would be a potent agent for the targeted ablation of HER2-positive PMGC. We thus investigated the therapeutic efficacy of a-RIT using 211At-tras in preclinical mouse models of HER2-positive PMGC
Material & methods
The in vivo biodistribution of 211At-tras was determined using both s.c. and PMGC xenograft mouse models. Astatine-211-labeled trastuzumab was injected into the tail vein (s.c. xenograft model) or the peritoneal cavity (both xenograft models). The PMGC mouse models were established by i.p. injecting luciferase-transfected N87/Luc cells (3 x 105) into 5-week-old B17/Icr-scid/scidJcl (homo) female mice 1 week before the experiment. These PMGC model mice then underwent RIT at Day 0, i.e, 1 week after cell inoculation. Mice received a single i.p. injection of PBS, Tras, non-carrier 211At (1 MBq), or 211At-Tras (0.1 or 1 MBq). Tumor growth in the PMGC mice was monitored every week using an in vivo bioluminescence imaging Fusion system (Vilber Lourmat, France). 10 min after the injection of luciferin, bioluminescence from PMGC was captured for 10 s. The total bioluminescence intensity in the abdominal region was quantified using Bio-1D software (Vilber Lourmat).
Results
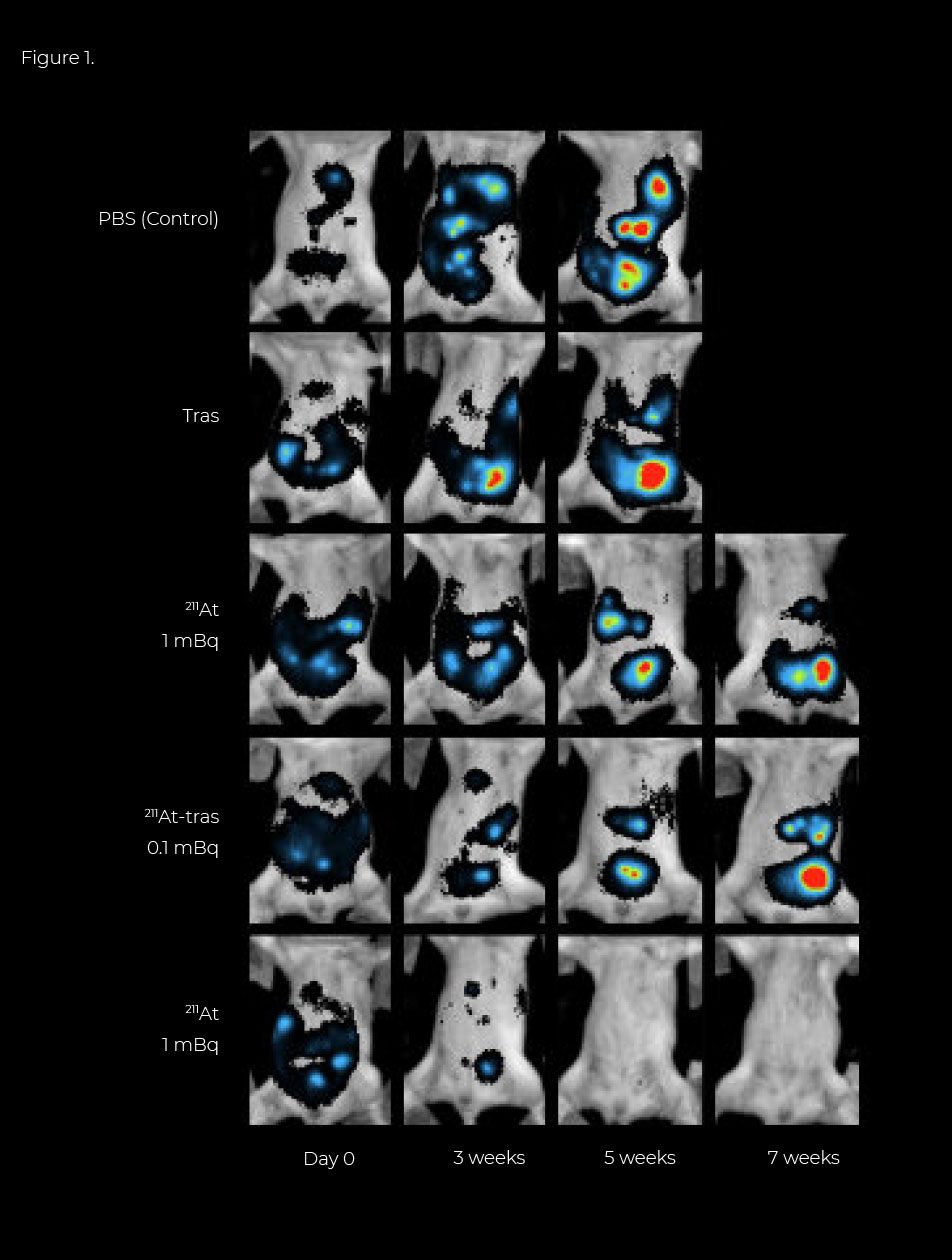
Figure 1. Locoregional therapy with astatine-211 labeled trastuzumab (211At-tras) in a mouse model of peritoneal metastasis of gastric cancer (PMGC) and tumor monitoring. The PMGC mice received a single i.p. injection of PBS, unlabeled Tras, free 211At (1 MBq), or 211At-tras (0.1 or 1 MBq) 8 days after N87/Luc implantation. The tumor growth was monitored longitudinally in each group by in vivo bioluminescence imaging (Fig. 1b). Representative bioluminescence images of the mice from each group are shown in Figure 1(b). Tumor growth was evident in the PMGC mice injected with PBS, unlabeled tras, free 211At, and 0.1 MBq 211At-tras. In contrast, a single injection of 1 MBq 211At-trastuzumab reduced the tumor burden at 3 weeks after injection, with the tumors becoming undetectable at 5–7 weeks after treatment. Trast and PBS injections had little to no effect on tumor growth in the PMGC model. Representative bioluminescence images of tumor growth in the mouse model treated with PBS (control), unlabeled Tras, 211At (1 MBq), or 211At-tras (0.1 or 1 MBq). Images were captured on the day before treatment (Day 0) or at 3, 5, or 7 weeks after treatment. The color scale indicates the luminescence intensity per pixel.
Results
In the current study, it has been demonstrated that 211At-trastuzumab specifically targets HER2-positive GC cells, both in vivo and in vitro and thus successfully inhibits tumor growth and improves survival in PMGC model mice. Biodistribution analysis in this PMGC mouse model revealed that the i.p. administration of 211At-trastuzumab (1 MBq) was a more efficient means of delivery of 211At into metastatic tumors than i.v. injection ; the maximum tumor uptake with i.p. administration was over 60% injected dose per gram of tissue (%ID/g) compared to approximately 18%ID/g with i.v. injection. Surprisingly, a single i.p. injection of 211At-tras (1 MBq) was sufficient to completely eradicate intraperitoneally disseminated HER2-positive GC xenografts in two of six treated mice by inducing DNA doublestrand breaks, and to drastically reduce the tumor burden in another three mice. Accordingly, locoregionally administered 211At-trastuzumab can specifically bind and effectively kill NCI-N87 (N87) cells, which are HER2-positive human metastatic GC cells, both in vitro and in a xenograft mouse model (s.c. tumors).