Chemiluminescence
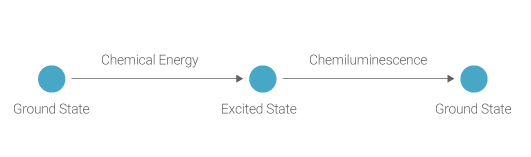
Chemiluminescence is the emission of light by a chemical reaction. It occurs when a chemical reagent containing stored energy releases light. The reagent is normally stable and does not emit light, but can be converted into a light emitting product, for example after interaction with a specific enzyme. In most contemporary ECL systems, the enzyme horseradish peroxidase (HRP) conjugated to a secondary antibody is the catalyst that fulfills this function.
The light produced is proportional to the amount of labeled compound in the sample and can be detected on X-ray films as well as by using CCD camera-based imagers such as the Fusion system. ECL is more versatile than general colorimetric methods as these antibody-based systems are usually designed to target specific biomolecules. In addition, the technique is fast and sensitive; signals are generated in seconds and relatively small quantities of antigens and antibodies are normally consumed. As light is generated without an external excitation source, there is no risk of photodamage to samples.
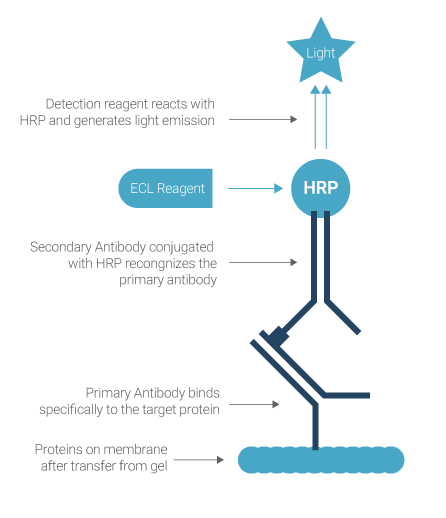
Chemiluminescence-based protein detection. The HRP conjugated secondary antibody will recognize the primary antibody directed against the protein of interest. This enzyme catalyzes the conversion of the ECL substrate into a sensitized reagent, which on further oxidation produces an excited state that emits light (428 nm) when it decays.
Sensitivity
The most recent ECL reagents from give rise to high sensitivity and linearity of signal response over a wide range of protein levels. It enables detection and precise quantitation of both high and low abundant proteins on the same blot after a single exposure.
Signal stability
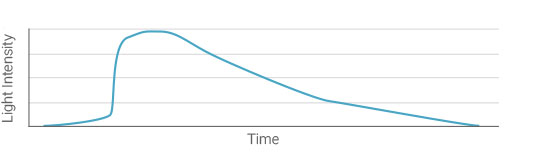
The light signal emitted by an ECL reagent reaches its maximum intensity shortly after activation and then diminishes with time. The rate of signal quenching varies among the available reagents but usually lies in the range of a few minutes to 2 h. This enables multiple exposures and a convenient time window between the end of the experiment and image capture.